Luck, foresight and science: How an unheralded team developed a COVID-19 vaccine in record time
Jennifer Haller layered a blue denim shirt over a gray tank top that morning, knowing she would later need to bare one shoulder. Her husband, always on her case about skipping breakfast, scrambled some eggs. The night before, she had read news online that the first person would get an experimental vaccine against the novel coronavirus at Kaiser Permanente in the morning. She wondered if that person might be her.
When she arrived at the research center, not far from Seattle’s iconic Space Needle, the whole world was standing by. It had been 75 days since news broke of unexplained cases of pneumonia seeming to emanate from a seafood market in Wuhan, China, and 66 days since scientists in the United States stared at the virus’s genetic code and vowed to conjure a vaccine to shut it down, at a record-shattering pace.
Doing so was a bet that, a few years ago, would have felt as audacious as sending a man to Mars. Vaccines took decades to develop. Their history was littered with missteps and disappointment. Jonas Salk’s polio vaccine came only after 50 years of trial and error. Vaccines for HIV and Zika still elude scientists.
Now the country was in the grip of a disease that had infected 172,000 people, killed 6,700 and was accelerating unimpeded. Dr. Anthony Fauci, who had quickly become the voice of the scientific community, was already predicting a vaccine within as little as a year, a projection many of his colleagues considered wildly optimistic.
But Fauci knew something many did not. Members of his team at the National Institutes of Health and an affiliated biotech company had been preparing for years for just this moment, just this vaccine. The liquid about to go into Haller’s arm was the culmination of years of research by a handful of scientists who, by virtue of incredible luck and incredible foresight, were prepared for one virus more than almost any other: a coronavirus.
Had this virus come 10 years earlier – or even five – science would not have been ready.
At the start of the pandemic, no one could have foreseen how controversial a COVID-19 vaccine would become – that the country would soon be stewing in anti-science and anti-vaccine sentiment, awash in misinformation and cleaved by mistrust.
When Haller had answered a callout from a friend to “Fellow Humans” to participate in a 14-month medical trial, she’d barely blinked. The 43-year-old mom had stepped up because it was the right thing to do in an awful moment.

As she clicked through the news on her phone and surveyed the medical staff and journalists surrounding her that morning, she was just figuring out that she was, in fact, making history. She was the first COVID-19 vaccine trial subject.
A pharmacist in an N95 mask, protective goggles and blue latex gloves held up a syringe labeled with an expiration a few hours away. The photographers focused their lenses on Haller’s shoulder. The needle slid in.
The experiment had begun.
Unsung scientists and the deaths of two infants
Then-President Donald Trump credited his Operation Warp Speed for the fast development of the vaccine, and marshaling the resources of public health agencies no doubt played a critical role. So did committing $9 billion for human trials and manufacturing the vaccine even before it was tested, banking on a green light from the U.S. Food and Drug Administration.
But the real credit for the rapid turnaround belongs to a series of uncelebrated discoveries dating back at least 15 years – and a constellation of unsung scientists.
Chief among them is Barney Graham, now 67, who had spent much of his career trying to understand why two infants died in a disastrous 1966 vaccine trial for a virus that remains the leading cause of hospitalization for young children. The tragedy slowed the development of a vaccine for respiratory syncytial virus (RSV) for decades, and it haunted Graham. He continued his research for 31 years until, in 2013, he had a major breakthrough – one that would become vital to developing a COVID-19 vaccine.
In recent years, as deputy director of the National Institutes of Health’s Vaccine Research Center, Graham’s mission had been to make a vaccine fast enough to stop a pandemic. He was looking for a good virus candidate.

A towering, gentle man with a medical degree and a doctorate in immunology, Graham had gotten close once before when the mosquito-borne Zika virus reached the U.S. in 2016. Thousands of babies were born with severely shrunken brains, a deformity known as microcephaly.
The Zika vaccine candidate held the speed record for getting from initial research into a clinical trial – roughly seven months – but that pandemic subsided before Graham could complete the small trial, leaving the world with no vaccine for Zika.
At the end of 2019, Graham was months into plans to make another vaccine at supersonic speeds. He had chosen a brutal virus called Nipah that inspired the 2011 film “Contagion.” Spread by bats, it last broke out in India in 2018, killing 17 of the 19 people it infected in Kerala.
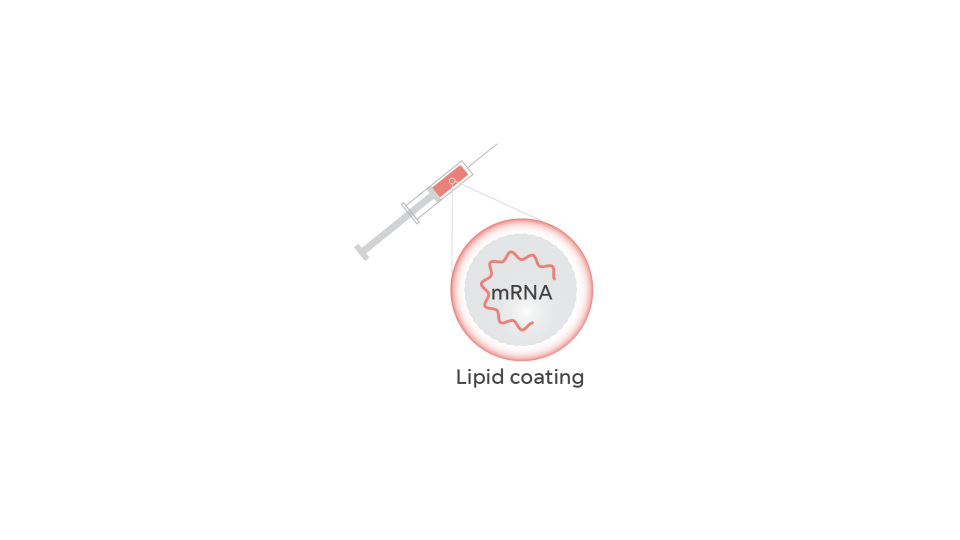
Though he's reflexively modest, Graham had a bold goal. He’d even picked a biotech company to make the Nipah vaccine: Moderna. During the Zika experiment, Moderna had developed a vaccine 10 times more potent than the DNA vaccine Graham himself had created. It relied on messenger RNA, sometimes called the “software of life” because it carries out the instructions of our DNA. In a vaccine, mRNA instructs our bodies to produce just a piece of the virus, the protein on its surface.
Moderna is so dedicated to this new world of medicine that its name combines the word “modified” with RNA. Its technology expedites development of vaccines by persuading the body’s own genetic process to mimic a component of the virus and create antibodies. Although in its decadelong existence Moderna had never brought a vaccine to market, it had tested several experimental vaccines in a dozen small clinical trials.
As Graham got word through back channels that the new virus in China was probably a coronavirus, he reached out to Moderna’s CEO, who was vacationing in France. We should scratch the Nipah plan, he urged Stephane Bancel in a Jan. 6 email, in favor of a different proof of concept related to the Wuhan outbreak.
“If it’s a SARS-like coronavirus, we know what to do,” Graham wrote. “This would be a great time to run the drill for how quickly can you have a scalable vaccine.”
Graham later laid out the idea for Fauci, his boss’s boss, in a conference room at NIH. Fauci is no micromanager; he hadn’t even been aware until then how confident Graham was in his ability to make a coronavirus vaccine.
There had been two other novel coronaviruses since 2003, although neither SARS nor MERS were terribly contagious and neither became pandemics. In early January, there was no reason to assume COVID-19 would be any different. Yet Graham already had his team diving into how to defeat the new coronavirus just to prove it could be done. Fauci was sold.
“Let’s go full-blown,” he said. “Let’s make a vaccine.”
Fauci had already set aside $5 million for the small Nipah demonstration project. Graham asked if there would there be millions more available.
“Barney, let me worry about the money,” Fauci replied.
If everything went perfectly, Graham said a vaccine could be ready within 12 to 18 months – the prediction Fauci would soon make public.
Jan. 10: The virus’s unique identity is revealed
Close to midnight on Jan. 10, Graham was relaxing at his suburban Maryland home when he finally got the news he’d been expecting: Chinese scientists had posted the genetic sequence for what they called the “Wuhan seafood market pneumonia virus.”
Early the next morning, Graham fired up his laptop in his home office, overlooking his front yard. He navigated to a website used by scientists worldwide to share information and saw hundreds of rows of four letters – a, c, g and t – that revealed the virus’s unique identity. The reports were true. It was a novel coronavirus, SARS-CoV-2, a new version of a family of infectious diseases.


According to news accounts, the virus so far had infected only 41 people and killed one. The story in The Washington Post about it that morning was buried inside the paper.
Graham began texting, emailing and calling a handful of close associates. He reached one, a former NIH scientist now at the University of Texas, Jason McLellan, just back from a Utah ski trip. Another was a rising star he had known since she was a teenager, when she applied for an internship. Kizzmekia Corbett had been working in Graham's lab since 2014, studying the best way to defeat coronaviruses with a vaccine.
The team began to tailor a vaccine to the new coronavirus. They had a big head start, having experimented on coronavirus vaccines in mice for years in collaboration with Moderna. The work happened on laptops, not in labs.
They moved fast, but carefully; the more accurate they were, the more effective the vaccine would be. It took them only a few hours.
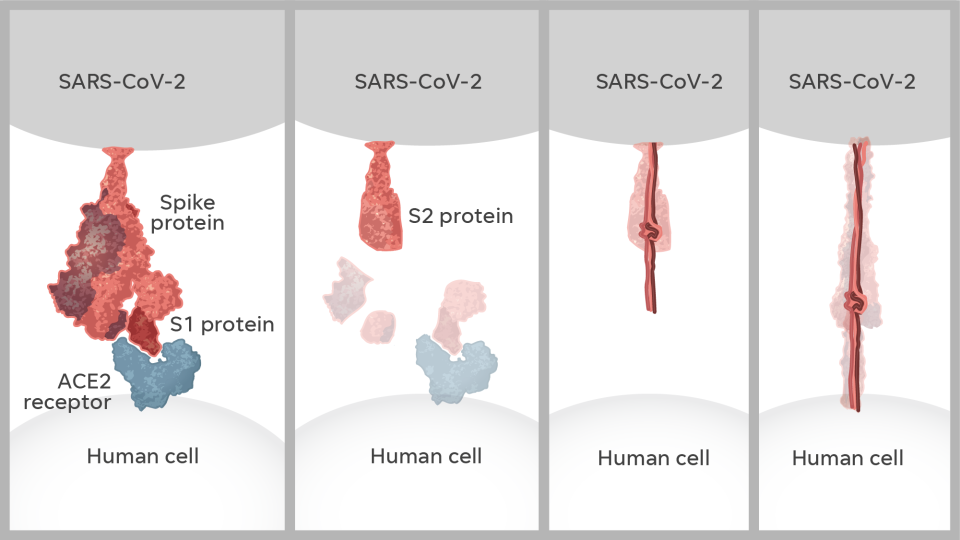
The mRNA would be encased in a fat bubble and injected in a person’s arm. It was intended to produce a protein identical to the spike protein on the surface of the new virus. That protein, they hoped, would trigger the body to produce just the right antibodies to fight back
Graham convened over the phone with scientists at Moderna two days later to sketch out the road map. Moderna would produce the vaccine, using the genetic code Graham provided. It would be the only vaccine for which the government would lead the first clinical trial, a trial Graham wanted to launch in a matter of weeks.
In Graham’s mind, he had clicked a stopwatch: The race to a vaccine had begun.
Jan. 19: The first U.S. patient is confirmed
Eight days after Graham’s team designed a vaccine, the new coronavirus officially reached the United States. In a Seattle suburb, a 35-year-old man who’d just returned from Wuhan began coughing and throwing up. He’d never gone to the seafood market where the virus was believed to have spread. After four days of symptoms, he called a local urgent care clinic for guidance on Jan. 19.
The clinic ran a batch of tests and found nothing. It overnighted the man’s nasal swab to the Centers for Disease Control and Prevention in Atlanta, gave him an N95 mask and told him to quarantine at home.
The next day, the man got a call. The test results had come back: He was the first patient in the United States confirmed to be infected with the new virus.
At the CDC’s request, Providence Regional Medical Center – which ran the urgent care center and had a hospital near Seattle in Everett – sprang into action. Using a protocol for Ebola, it sent emergency medical technicians to pick up the infected man. The techs wore hazmat suits. They used a gurney equipped with an Iso-Pod, a plastic bubble to keep the virus from spreading.
When they arrived at the hospital, they wheeled him to the biocontainment unit, with a special air flow system to prevent the virus from escaping his sealed room. Doctors examined the man via a video camera.

The patient didn’t feel too sick – at first. He took his own temperature and barely had a fever. Later he would develop pneumonia, from which he would recover.
Local public health officials scrambled to track down at least 50 people who’d had contact with the man in the five days since his return from China. They never found anyone sick, leaving them unsure how contagious this new disease might be. The Seattle Times quoted health experts saying not to worry too much; coronaviruses aren’t usually that contagious until someone has symptoms.
From Davos, Switzerland, a CNBC correspondent asked Trump on Jan. 22 if he was concerned about a pandemic.
“No, we’re not at all. We have it totally under control. It’s one person coming in from China,” Trump said, referring to the patient in Seattle. “It’s going to be just fine.”
Although it still wasn’t clear how urgent a vaccine would become, Moderna was already starting to produce the one Graham and his colleagues had developed. NIH put out a notice to 10 sites it uses for vaccine trials, asking them to get ready to test the experimental concoction on humans.
On Jan. 27, a year ago today, Dr. Lisa Jackson at Kaiser Permanente Washington in Seattle learned that she would oversee that trial. Jackson dropped all her other work to focus on the vaccine.
Feb. 19: Results from mouse studies hold promise
Experts knew there had never been a vaccine created in less than four years. That was for the mumps, and even that vaccine was an outlier. “I think the goal of 18 months is one that will be very, very difficult to achieve,” Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, said in the early months. “But it just may be our moon shot.”
By Feb. 19, Graham and his team had the first good news from a two-week mouse study. Blood drawn from vaccinated mice showed they were producing antibodies to fight the virus. Corbett designed that study, gave mice the vaccine and analyzed the results. It was second nature for her, something perfected over years studying other coronaviruses.
For Graham, the mouse results were simply reaffirmation that he was right to be so sure of the vaccine, confidence earned by devoting so much of his life to trying to develop a vaccine for RSV.
Graham’s high school years in the late 1960s were spent on a farm near a speck of a town, Paola, Kansas, where he helped his father, a dentist, raise quarter horses, cattle and hogs. After medical school and a doctorate, Graham had spent much of his career at Vanderbilt University, where he focused on HIV as well as RSV. He brought that work with him to NIH when he was recruited there in 2000.
RSV sends as many as 125,000 children to the hospital each year. It also holds a special place in the history of vaccines. In one clinical trial in 1966, 31 infants were given the trial vaccine. Twenty of them later caught the disease. Of those, 16 ended up in the hospital and two died.
McLellan, a young postdoctoral fellow at NIH when he began working with Graham, says his mentor was devastated to see children with RSV struggling to breathe. Starting in 2009, Graham worked shoulder to shoulder with McLellan, fresh from earning a doctorate in biophysics from Johns Hopkins University. Together, they were going to solve the riddle of how to make a safe vaccine for RSV.
With some vaccines, such as the one Salk created for polio, scientists took a live virus and killed it. The dead virus went into the syringe, then into arms. It was imprecise science and, in the case of polio, hugely effective.
That approach did not work for RSV; the dead virus made children more vulnerable to the illness. Graham suspected that to unravel the situation, he needed to understand precisely how viruses attack human cells, something NIH had been studying with HIV.
Proteins on the surface of a virus attach themselves to a cell membrane, tearing open a pathway for the virus to invade the cell. Graham wanted to understand how the protein accomplished its mission. McLellan wasn’t sure how useful this pursuit would be. It hadn’t worked for HIV. He didn’t know whether it was a bad idea or whether HIV was just too daunting of a virus.
They wanted to see for the first time what the protein on an RSV virus looked like. McLellan used an antibody, isolated from a human, to pin the protein and make it stable. Using an advanced technique known as X-ray crystallography, he then froze and X-rayed it.
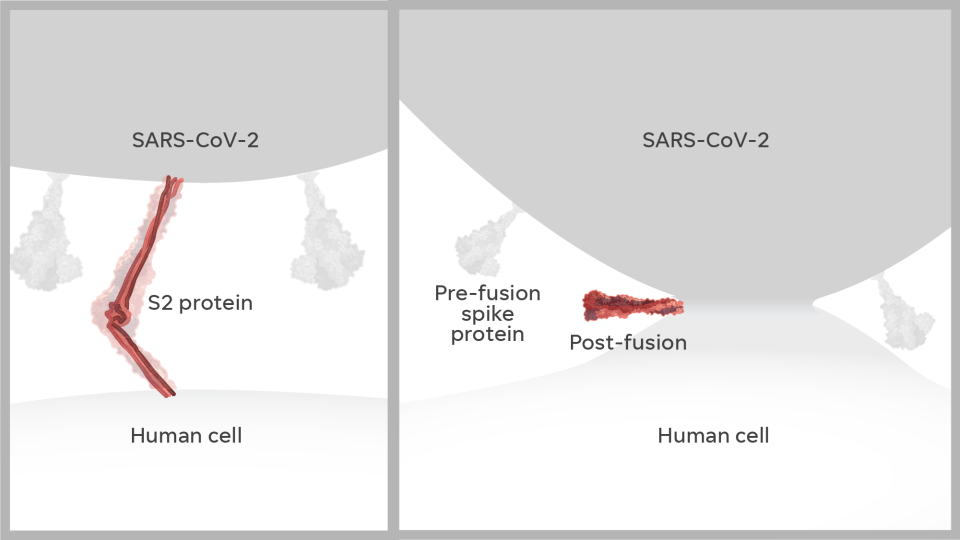
As he sat next to Graham at a scientific event in China, McLellan pulled up the image. It looked nothing at all like Graham had expected. It turned out that a protein looks different after it attacks a cell – before, it’s like a lollipop; after, it’s like a golf tee.


When surface proteins bind with a cell, it resembles the attack of the creature in “Alien.” A peptide shoots out and latches onto cells, then rips the cell membrane apart, giving the virus access to the cell. Our immune systems produce different antibodies for different shapes. The wrong antibody offers less protection – and could even make the virus worse.
Graham recognized immediately that this level of precision was a game changer. The research is now being used to develop an RSV vaccine, the vaccine Graham had thought might be his legacy. Instead, when MERS broke out in Saudi Arabia in September 2012, his team, including McLellan, immediately began researching the structures of proteins on coronaviruses.
McLellan continued to work with Graham to identify the spike protein on coronaviruses, even after he moved on to Dartmouth College and later the University of Texas. By 2017, they had figured out how to defeat coronaviruses with a vaccine and published a major scientific article, one that nearly all vaccine makers would one day rely on for COVID-19.
Subsequently, Corbett at NIH worked with others to test possible vaccines, with a special focus on MERS, also known as camel flu. Working with Moderna, she helped inject mice, then drew blood to test for antibodies.
That preparation left them ready when another novel coronavirus appeared.
Feb. 28: A Seattle doctor gets troubling news
Back in Seattle, the first human trial for the coronavirus vaccine was about to start. A process that typically takes months – approving a study design – became Jackson’s sole priority and was completed in days. The plan was to give different doses to four groups of 15 adults, ages 18 to 65. Two shots would be given 28 days apart, with months of follow-up.
The point was to see whether the vaccine had side effects, begin to settle on its proper dose and determine whether it was producing neutralizing antibodies to stop a virus.
At Kaiser Permanente, Jackson was hiring nurses and getting supplies ready when bad news struck at a suburban hospital on the other side of Lake Washington from her downtown research office.
Quite a few patients at Kirkland’s EvergreenHealth Hospital had pneumonia. As medical director of infectious disease, Dr. Francis Riedo knew this wasn’t unusual during the winter but, since the CDC had just loosened the criteria for coronavirus testing, Riedo decided to test two of the patients. He chose them based on which were in hospital rooms that could easily be sealed off if the results came back positive.
Swabs were collected that night and sent to the local public health office. The next morning, Riedo was shocked by the results: Both patients had COVID-19.
One – a man in his 50s who had never left the country – died that same day. The other, who came from a nearby nursing home, died later. The two patients had no connection.

Riedo realized that the virus had been circulating in the Seattle area for weeks. The epidemic, he concluded, was already out of control. Soon, the rest of the nation would come to the same realization.
Two residents of a nearby nursing home, Life Care Center of Kirkland, also tested positive. Fifty more were awaiting results. At a standing-room-only press conference the next day, Riedo predicted: “What we’re seeing is the tip of the iceberg.”
Overnight, Seattle became the nation’s first hot spot. The CDC flew in experts. A local school district decided to shut down. Gov. Jay Inslee declared a public health emergency. Within a few days, much of the rest of the country would go into a lockdown.
March 3: Jennifer Haller volunteers for the vaccine trial
Amid this frenzy, Jennifer Haller, an operations manager at a Seattle tech company and a mother of two, started paying close attention. Her mother and stepfather lived not far from the nursing home, and Haller was worried about them.
On March 3, she was scrolling through Facebook at work when a friend’s post stopped her. He had linked to Kaiser’s signup page as a favor for a nurse working on the vaccine. This was the Phase 1 trial that Graham, now exclusively working from home, had set in motion and was helping oversee.
Haller didn’t care about the $1,100 stipend being offered to trial participants. She had always felt that as a white person living a middle-class life, she should give something back. She also was game for taking risks. At a previous job with a stun gun company, she had volunteered to be struck so she would know how it felt. It was a shock she hopes to never feel again.
So, she clicked on the link and filled out a form that asked only one question aside from her age and basic contact information: “Are you willing to attend 11 in-person study visits and have 4 phone visits over a 14-month period?” She clicked submit.

Haller put it out of her mind until two days later, when her phone rang just as she sat down in a restaurant with a friend. She almost never answers calls from strangers. This time she did, and it was someone from Kaiser asking if she had 20 minutes to spare. She excused herself from the table to answer questions about her health history and whether she could make all the appointments.
From there, things moved quickly. Haller went into Kaiser’s offices a couple of days later for blood draws, a physical exam and more questions.
Because of so much earlier research, led by Graham and Moderna, no one involved expected the vaccine to make the disease worse. Still, side effects are common in vaccine trials, including pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, swollen lymph nodes, nausea and vomiting, and fever.
If any serious side effects emerged that weren’t expected, the trial would be immediately suspended – even halted for good.
Haller knew she was essentially a guinea pig. It didn’t faze her.
March 16: Haller receives the first U.S. vaccine
As the coronavirus outbreak in Seattle grew, officials at NIH feared the crisis could shut down the vaccine trial at Kaiser. They went back through the list of potential sites and added Emory University in Atlanta as a secondary hub on March 12.
Emory scrambled to put together a protocol. The Atlanta trial would be led there by Drs. Nadine Rouphael and Evan Anderson, both veterans of infectious diseases and vaccines.
Kaiser told Haller to report to its downtown Seattle office the morning of March 16 and asked her if she would be willing to have the Associated Press there to record it. No big deal. She signed the waiver.
When she walked into the downtown office, she still had little clue how much attention she was about to get. After the shot, she was asked to wait a couple of hours just in case there were any reactions. She felt fine.


Shortly before Haller walked out, notifications on her cellphone started going off. The first text came from her mother. “Just received a call from Deborah Horne at KIRO TV. … They would like to talk with you and want you to call.”
Hours later she was live on CNN and MSNBC.
“I’m excited to be the first person,” Haller told MSNBC’s Ari Melber. “This is crazy.”
“Crazy is one word for it,” Melber joked, making Haller laugh.
Haller’s shot marked an enormous milestone for Graham and his team. It had been only 66 days since the Chinese posted the genetic sequence. They had proven that vaccines can be ready for human tests quickly. Graham remained fully confident in the vaccine, later confiding to a reporter that at that point he already expected it to be about 80% effective.
If he turned out to be right, expectations about the speed of vaccine development would change forever.
May 9: The first results of the trial appear
In Atlanta, another snag cropped up. A courier service was trying to deliver vaccines for the Emory leg of the trial from a repository at Fisher BioServices in Germantown, Maryland. However, flights from the nation’s capital kept getting canceled as the country was finally shutting down. That was a problem, because the vaccine solution would degrade if it was not kept frigidly cold.
After two days of delays at Reagan National Airport near Washington, D.C., a courier service that specializes in shipping medical products gave up on flights and used its own delivery vehicles to drive down to Atlanta instead. It packed the vaccine containers in dry ice, dropping them off late the evening of March 26.
The first shot was given there the next morning – nearly two weeks after Haller had kicked off the Phase 1 trial.
Anderson worked from early morning to late at night managing the trial. A key part of the analysis, being done largely at Vanderbilt University, was to test the subjects’ blood samples for antibodies that would block the virus. That data was discussed in regular conference calls with Graham and the team at NIH, along with others involved in analyzing the data.
Accounts vary on exactly when the researchers knew the trial was a success. Anderson remembers seeing the data on his computer; Jackson remembers hearing about it in a conference call.
Graham pegs it to an email from researchers at Vanderbilt on May 9. They had the results of antibodies examined from the first eight volunteers, including Haller. The tall S curves on the chart told the story – the higher the top of the S, the better. When Vanderbilt researchers took antibodies from the volunteers’ blood and tested them on infected cells in the laboratory, the virus stopped replicating.
Graham was expecting the vaccine to produce neutralizing antibodies, but not this strong. The highest dose tested was dropped because it produced the most side effects, including fever, but otherwise there were no major safety concerns.
In a conference call with all the key players, the team gave each other verbal high fives – “Wow!” and “Thank you!” – for a job well done.
On May 18, Moderna made the unusual decision to release those early findings in a press release. The company’s stock soared that day, up 250% since December. Haller figured out she probably was one of the eight volunteers studied – and was pleased that the vaccine seemed to work.
Within days, Moderna announced plans for a 600-volunteer Phase 2 trial to establish a dose. That trial was barely underway when the company started crafting the Phase 3 study, which would balloon to 30,000 participants, using the most promising dose from Phase 1 instead. Time was of the essence.
They believed the vaccine was safe; now they had to see whether it worked.
April 1: Dolly Parton steps up with $1 million
By the end of March, the pandemic looked grim. The nation was in lockdown, nearly 200,000 cases had been confirmed in the United States, and the World Health Organization was estimating that without a vaccine at least 2 million Americans could die of the disease.
Country music legend Dolly Parton posted a hopeful video on Instagram for her 3.8 million followers. “Hello, this is Dolly climbing the Stairway to Heaven because this virus is scaring the H-E-L-L out of us,” she sang, adding that she thought God was testing us and “when this passes we’re all going to be better people.”
Then, on April 1, she again took to the social media platform to announce that she had donated $1 million to her hometown school, Vanderbilt University, to help find a cure for coronavirus. Parton had bonded with one doctor in particular there after she was injured in a car accident. Months later she would learn along with the public that some of her money went to Moderna’s Phase 1 study, a fact revealed when findings were published in the New England Journal of Medicine.
One of the challenges of making a vaccine is money. Not all viruses become epidemics, reducing the public pressure – and funding – to stop them. And developing a vaccine involves enlisting thousands of volunteers who are healthy, and then waiting to see how many get infected with the virus. These trials are expensive, and pharmaceutical companies don’t always reap much profit at the end of it.
If a COVID-19 vaccine was going to be available as soon as possible, the U.S. government was going to have to open the Treasury to pay for research, buy doses in advance and get the manufacturing started on spec, well before the vaccines were even up for approval. It did. Two weeks after Parton made her Instagram news, on April 16, the Department of Health and Human Services pledged to spend $483 million on the Moderna vaccine. Later, it would add another $472 million to pay for the trial.
Other vaccines in development could save millions of lives each year worldwide with similar infusions of funds. When asked why so much money was being poured into a COVID-19 vaccine, Corbett, one of the leaders of vaccine development at NIH, said, “I don’t really have an answer other than people are dying. And also, frankly, people in the United States are dying, which changes a lot.”
Goldman Sachs estimates that Moderna will make $13.2 billion in revenue this year from the vaccine.
July 27: Moderna’s Phase 3 trial begins
“It’s a big day: It’s the first Phase 3 of a COVID vaccine in the U.S.; it’s the first Phase 3 of an mRNA vaccine ever; and it’s the company’s first Phase 3 as well,” Moderna CEO Bancel told CNBC’s Squawk Box on July 27. “So a big day came from a lot of work of a big team.”
Having fully taken the reins from Graham and his colleagues, the company now needed to recruit 30,000 people – a massive number set by the FDA – which Bancel estimated would take up to eight weeks. Infection rates would determine when they would have results; the worse the outbreak, the faster they would have data showing whether the vaccine worked.
“October, maybe November,” Bancel said. “It’s tough to know right now.”

Bancel had been CEO since October 2010, recruited by venture capital firm Flagship Ventures to run the company it recently had funded. A chemical engineer with an MBA from Harvard, Bancel had held the top job at a French biotechnology company, BioMérieux, at 36 and pushed the company to dramatically increase its market share. Bancel flew to Boston and was intrigued by the idea of leading a company that could revolutionize medical science. He talked to his wife about it and said, “Look, this could change the world.”
Early employees describe Bancel as focused on filing patents. Once he realized there were 22,000 proteins in the human body, one employee said, he wanted his scientists to produce as many as possible – regardless of their scientific significance – to corner patents on RNA technology. In meetings, Bancel would stand before a whiteboard and often curse at his staff. Resignations, firings and poor reviews on Glassdoor followed.
Bancel was an excellent evangelist for the company, though. In a 2013 TED talk, he predicted based on mouse studies that RNA could regrow damaged heart tissue after a cardiac arrest – research that Moderna is working on today in collaboration with AstraZeneca.
The message was clear: Moderna intended to revolutionize medical science.
Vaccine relies on several key breakthroughs
It was not a given when Moderna was founded that it would ever be at the forefront of a pandemic vaccine. While much has been made of the messenger RNA technology, the two companies trying to use it to stop COVID-19 – Moderna as well as BioNTech with help from Pfizer – only turned their attention to vaccines in the past five years or so.
Moderna owes its existence to a Harvard professor, Derrick Rossi. Both Moderna and BioNTech also were built upon the discoveries of two researchers at the University of Pennsylvania, Kaitlin Kariko and Drew Weissman.
The stories of the three academic scientists weave together. But while Kariko now works for BioNTech and Weissman is consulting for the company, Rossi has largely been written out of Moderna’s history.

Most advances in science are incremental. Rossi’s breakthrough, trumpeted by Time magazine as an advancement that could one day cure intractable diseases, built on a truly startling discovery.
On June 30, 2006, Shinya Yamanaka, a doctor and researcher at Japan’s Kyoto University, had given a presentation at a Toronto conference that stunned a roomful of scientists. Speaking quietly, frequently looking down at his notes, Yamanaka explained that he had been able to take mature cells from mice and convert them back to embryonic stem cells. It was akin to biological time travel.
Yamanaka was describing a discovery that might allow scientists to take cells from one part of the body, such as the skin, convert them back to embryonic cells and then coax them into becoming some other type of cell.
His discovery had dramatic implications for future transplant patients, whose lives can depend on waiting for a perfect donor. Yamanaka was describing a method that might allow someone with end-stage liver disease to instead have a new liver grown from cells containing their own DNA.
Rossi was among those in the room, marveling at how elegant and awe-inspiring it all seemed. Six years later, Yamanaka would take the stage in Oslo, Norway, to accept a Nobel Prize.
Yamanaka’s method was highly inefficient, however, and the method he used could embed code in a person’s DNA and possibly cause cancer. Within a year of the Kyoto presentation, Rossi dove into Yamanaka’s work to find a solution for its biggest drawback: the use of retroviruses.
By then Rossi was working at Harvard’s Immune Disease Institute, where one of his postdoctoral fellows, Luigi Warren, had started to consider what would happen if instead of using a retrovirus to take a cell back in time, you used RNA.
We all know DNA is the secret to life. It determines whether we’re redheads or brunettes, tall or short. More importantly, day by day and minute by minute, DNA transmits the genetic signals that keep our bodies functioning. Need insulin to keep your blood sugar in check? DNA sends the signal to produce insulin. Need an antibody to fight off a virus? DNA takes care of that.
DNA sends those signals by making RNA, which delivers DNA’s messages to cells. Genetic scientists have a saying: DNA makes RNA makes proteins makes life.
In essence, Warren’s theory was that RNA could instruct our bodies to produce the drugs needed to fight disease or to inoculate ourselves against viruses.

One day, Warren put RNA onto mouse cells in a petri dish to see if he could get them to produce protein. He opted for a protein that glows fluorescent green so it would be easy to tell if the process worked. After a while, Warren looked into the microscope. His heart beat faster. He rushed over to Rossi, urging him to come take a look.
As Rossi peered into the microscope, he couldn’t believe what he saw: The cells were glowing green. Cells from mice had produced a protein that doesn’t even exist in mice. RNA worked.
That day in 2009, Warren and Rossi thought they had found the solution – one that exposed additional problems. Making the conversion back to embryonic cells takes time; two weeks in mice, to be exact. RNA is notoriously unstable. To keep the proteins glowing, Warren was going to have to add more RNA each day. Every day, the cells grew dimmer, then stopped making the protein – and died.
The men were stumped. It dawned on Rossi that others had probably already tried this, which led him to the two scientists at the University of Pennsylvania, who had been working on RNA for more than a decade with the same challenges. When they gave RNA to mice, the mice hunched up and refused to eat. Some died.
Weissman and Kariko had figured out that RNA would cause cells to become inflamed and reject the RNA as a foreign invader. They started toying with the idea of modifying the RNA to disguise it, fooling the immune response. They had been publishing about their successes for years.
Armed with their stealth method, Warren could keep his cells glowing for days. He also managed to convert the cells back to their embryonic state, an idea Rossi pitched to Flagship Ventures of Cambridge in 2010, starting the company today known as Moderna.
It would be several more years before Moderna began using the mRNA science to pursue vaccines. By then, Rossi was no longer with the company.
Sept. 7: Trump says, “We’re going to have a vaccine very soon”
On Aug. 6, trailing in the polls to Joe Biden, Trump suggested that the COVID-19 vaccine might be ready before Election Day. Although he had toured the NIH lab with Fauci, Graham, Corbett and others in March, the president had previously demurred to the experts on the timing of a vaccine, focusing more on rapid tests and treatments. Outside the White House, a reporter asked if a vaccine would help him win.
“It wouldn’t hurt,” Trump said. “But I’m doing it not for the election; I want to save a lot of lives.”
Critics jumped in, worried the vaccine was being rushed for political gain. A poll taken by CNN days later showed Americans were nervous. Only 56% said they would get the vaccine when it was approved; 40% said they wouldn’t. It didn’t help that the administration had called the program to accelerate development and production “Operation Warp Speed.”
Within a month of Trump’s comments, AstraZeneca temporarily halted its trial because of a single case of spinal cord inflammation. Researchers would later determine the case was not vaccine related, but it sent shockwaves through the public.
As time went on and coronavirus deaths continued to mount, Trump started taking shots at FDA and CDC officials for playing politics by delaying the vaccine.
“So we’re going to have a vaccine very soon, maybe even before a very special date. You know what date I’m talking about,” Trump said on Sept. 7.
Two days later, nine vaccine makers came together to respond, pledging in a joint statement to follow “high ethical standards and sound scientific principles” and to go through the normal channels of review by the FDA.
“We believe this pledge will help ensure public confidence in the rigorous scientific and regulatory process by which COVID-19 vaccines are evaluated and may ultimately be approved,” the statement concluded.
Looking back, Dr. Gregory Glynn, research and development president at Norovax, one of the nine companies, said, “It’s really important we give gold standard evidence.”
Although Graham was not involved in the public statement, he knew that the vaccine makers had no choice. Science has to proceed through prescribed steps, even when lives are in the balance. The FDA could not green-light any vaccine until safety and efficacy data was available and reviewed.
Sept. 24: Concern about lack of diversity in trial addressed
Not long after Moderna began recruiting volunteers for its final trial, the head of vaccine development for Operation Warp Speed noticed a big problem: The company wasn’t recruiting enough African Americans. Moncef Slaoui got on the phone with Bancel and Moderna President Stephen Hoge. When Slaoui, a former board member at Moderna, felt that Bancel wasn’t listening, he began shouting.
“We were shouting at each other on the phone – shouting in a respectful but very angry way, very stressed way,” Slaoui said. “There was a very big tension because we need to recruit very quickly and we need to recruit the right people.”
To Slaoui, it was clear that having Black and Latino participants in the trial was key to the vaccine’s future success. The virus was disproportionately lethal for those communities; African Americans die of COVID-19 at nearly three times the rate of white Americans. It also was important to show that the vaccine was safe for all, a detail often neglected in clinical trials.

Moderna relented and slowed enrollment for a while. Its recruiter bought social media ads targeted to Black and Latino users. Slaoui joined a virtual town hall meeting with the Rainbow/PUSH Coalition, headed by civil rights icon Jesse Jackson.
“Frankly, developing a vaccine not used in the population or in a fraction of the population is the same as having no vaccine,” Slaoui said at the Sept. 24 meeting. “A vaccine on the shelf is absolutely useless.”
Jackson blames the reluctance by Black and Latino people to participate on “racism in health care down through the years” in the United States, ranging from the Tuskegee experiment to poorer hospital systems in minority areas. He’s working to help overcome that skepticism.
“The history must inform us,” he said, “not paralyze us.”
The pause to recruit additional participants of color lasted two weeks, and resulted in a 50% increase in Black people in the trial. It also caused Moderna to lose its front-runner status to another company, Pfizer.
Nov. 16: Effectiveness of Moderna’s vaccine announced
Graham’s boss called with astonishing news on the second Sunday in November. The government’s data monitoring safety board – the only group allowed to see behind the curtain and know who in the trial received a vaccine or a saltwater placebo – had completed the first analysis of 94 volunteers who had gotten COVID-19 in the Pfizer vaccine trial. To meet the FDA threshold in the first analysis with only 32 subjects, the vaccine would have to be at least 75% effective.
The data showed it had far exceeded that threshold: It was more than 90% effective.
As Graham hung up the phone, he took a deep breath and told his wife that the Pfizer vaccine – nearly identical to Moderna’s – worked. Then he wandered over to his desk, sat down and wept tears of relief. The type of vaccine he had been perfecting over a decade was far more successful than even he had imagined.
Graham did not yet have any word on the Moderna vaccine. But Fauci did. He had been clued in days earlier by the head of the data safety board but had promised to keep the secret until Nov. 16.
When that day came, Moderna made a nearly identical announcement: Its vaccine was 94% effective.
Jonas Salk became an overnight hero on April 12, 1955, when the news was delivered in a University of Michigan lecture hall that the polio vaccine was 90% effective. With the COVID-19 vaccine, Graham and his colleagues are not yet household names.
Graham is unconcerned about that reality, preferring the laboratory to the limelight. Corbett finds it more difficult to hide her emotions, at least on Twitter, where her profile reads “Virology. Vaccinology. Vagina-ology. Vino-ology. My tweets are my own. My science is the world’s.” Replying to her own tweet the day Haller was given the first vaccine in an NIH trial, Corbett had written, “I.... I... This is... *tears*”:

The team was recognized on the first day of December with a scientific award, the brainchild of U.S. Rep. Jim Cooper, a Democrat from Nashville, Tennessee. Cooper sought to counter the Golden Fleece Award, which singles out silly-sounding research efforts, by pinpointing the research behind major breakthroughs. The 2020 Golden Goose Award went to Graham, Corbett, McLellan and others for leading the effort to create a vaccine.
Corbett responded with more tweets, one aimed at colleague McLellan: “Thanks Jason! We did it!!!” She also tweeted an inscription in a book given to her by Graham in 2009, when she was an NIH intern. It said, “I am also proud of how you have matured scientifically and professionally, and have every confidence that you will achieve your dreams and be very successful.”
In another video post, Corbett pauses at times to hold back tears, saying: “Part of the reason why I was given essentially the wings to fly with this project is because I had a mentor who allowed me to use his resources and sit in the back of his laboratory and to ask scientific questions that a lot of other people didn’t really care about.”
Dec. 17: Moderna seeks FDA green light
Then, it was showtime. Moderna’s top scientific leaders went before a panel of 21 independent scientists on an FDA advisory panel to present their data. The Dec. 17 meeting was held remotely via webcast – and the whole world could watch.
Their delivery was deadpan, even though this was the moment of truth. The panel’s recommendation would weigh heavily in whether the FDA would agree to an emergency use authorization.
Typically, the FDA spends months analyzing data to approve a vaccine. This time, that work was done in 2½ weeks, partly because the vaccine was so extraordinarily effective. In advance, the FDA had said it was willing to give the green light to a vaccine that was 50% effective. It was even willing to stop the trial early if the vaccine proved to be 75% effective. Moderna’s data, like Pfizer's before it, was even more dramatic.
Of the 196 trial participants who had COVID-19 with symptoms, only 11 had received the vaccine. All 30 cases of severe COVID-19 happened among those who got the placebo.
Subscribe: Help support quality journalism like this.
Later in the afternoon, an FDA doctor carefully walked the independent scientists through the side effects and adverse events. She concluded that while there were a few cases of severe side effects, such as deep muscle pain, she found no safety concerns in the data. Six people in the vaccine group had died, but none of those deaths – ranging from a heart attack to suicide – was considered vaccine related.
At the end of the day, the biggest question was whether to make it clearer that the group was voting on emergency use authorization and not regular FDA approval. In the end, no change was made to the language: “Based on the totality of scientific evidence available, do the benefits of the Moderna COVID-19 Vaccine outweigh the risks for use in individuals 18 years of age and older?”
The vote was 20 yes, with one abstention – an objection to the wording, not the vaccine.
Jan. 7: Vaccinations reach nursing home residents
Employees of the Ararat Home in Los Angeles buzzed with nervous energy as they queued up in the dining room on Jan. 7. They tried to stay 6 feet from each other, and all wore N95 masks. Some crossed themselves in preparation.
All eyes were on a Los Angeles County health official in a plastic robe and face shield. He and his colleagues were carefully setting up with hand sanitizer and wipes, gloves, needles and a sharps box, every so often jotting something down in their notebooks.
When he was finally ready, Susan Veranyan, a director at the skilled nursing facility, looked at him expectantly.
“Where is the vaccine?” he asked her gently.
Realizing that she had forgotten the main attraction, Veranyan ran back to an office mini-fridge. She pulled out a small, blue-lined box marked in red type: “Moderna COVID-19 Vaccine.” She’d start with 100 doses.

The rollout of the vaccine has been somewhat rocky. Pfizer, first out of the chute, said it didn’t get clear directions on where to send vials. Then states received the vaccines but didn’t have a coherent plan for giving out shots. In California, an increase in allergic reactions involving 10 people at one location led state health officials to temporarily stop using one lot of the Moderna vaccine. State officials later agreed the batch was safe.
Things went smoothly at the Ararat Home. As a nurse traveled from room to room, old ladies rolled sweater sleeves up their biceps like tough guys. One of them, Siranouche Haladjian from Russia, said she’s been afraid of contracting the virus because of her severe asthma. Her Lebanon-born roommate, Ayrik Ellyin, doesn’t speak English but knew the word to describe how she’s felt this past year: “Jail, jail, jail.”

Tamarah Keshishzadeh, who nearly died of the virus last year, asked one of the nurses to put lipstick on her before she received the vaccine. After being injected, Keshishzadeh started singing a low, Armenian warble while the nurse swayed alongside her.
“It’s a song,” Veranyan explained, “about old-days’ love.”
Epilogue
Fauci now attributes the vaccine being ready so quickly to two factors, one of them unfortunate. With the coronavirus spreading rapidly during the trial, odds dramatically increased that some of the 30,000 placebo recipients in the Moderna and Pfizer trials combined were going to catch the virus quickly. That would prove that the vaccine worked in the other 30,000 who received it.
The other reason, Fauci says, was the work Graham and his colleagues had been doing for years, in their own laboratories as well as in embracing mRNA and choosing it for the COVID-19 vaccine.
"I think the perspective that I had,” Fauci said, “was seeing the link from the years of fundamental basic and clinical research that got us to that point, in the first week of January, when we knew that all we needed was the sequence of the new coronavirus.”

More COVID-19 vaccines are underway. Other than one developed by AstraZeneca in partnership with Oxford University, all relied on the scientific breakthroughs coming from Graham and his colleagues. “As politically correct as I can say it,” Corbett said of the other major vaccine makers, “they’re catching up because they are copying and pasting.”
Back in the lab, McLellan has found a slightly different configuration of the protein that is easier to produce and more precise. It also appears to be more potent. Moderna and Pfizer both are reportedly exploring a next-generation vaccine that may require only one small dose, which might be ready in time to benefit poorer countries.
The battle to stop pandemics is not over. Another one is coming and it could even be another coronavirus. Currently thousands of known strains exist in bats that could jump to humans at any time.
Graham believes we’re now better prepared.
“I think with all the things that have been done during this outbreak, we would know how to make a new coronavirus vaccine once we had a new sequence,” he said. “And I think a lot of clinical testing, experimentation that’s going on now would be able to be bypassed and you’d be able to really deliver vaccine much quicker.”
Beyond that, Graham already has set his sights on the next big battle in the war against viruses: developing a universal vaccine to protect us against any coronavirus.
Next time, Graham wants to stop it before it starts.
Credits
Graphics: Jennifer Borresen, Ramon Padilla, Javier Zarracina and Mitchell Thorson
Photo editing: Chris Powers
Digital production: Mara Corbett
Contributing: Karen Weintraub
Deliver a safe, effective COVID-19 vaccine in less than a year? Impossible. Meet Moncef Slaoui.
The sprint to create a COVID-19 vaccine started in January. The finish line awaits.
This article originally appeared on USA TODAY: COVID-19: The inside story of the fastest vaccine ever developed

 Yahoo Movies
Yahoo Movies 
