COVID-19 vaccine rollout hasn't worked, but change is coming, vaccine panel predicts
The federal government needs to learn from Disneyland, bank teller lines and airline reservation systems, USA TODAY's panel of vaccine experts said.
Nearly unanimous in condemning the messy COVID-19 vaccine distribution rollout so far, the group also expects the operation will improve under the Biden administration.
What has been lacking, nearly all said, is strong leadership at the national level.
"Federal leadership failed to prepare fully to neutralize this pandemic. They concentrated on the vaccine with much less regard to vaccination," said Dr. Kelly Moore, deputy director of the nonprofit Immunization Action Coalition. "There’s a need for stability, reliability, and a process people can trust."
Improved direction, funding, coordination and communication – which Biden has promised to provide – should solve the problem of getting vaccine from pharmacy freezers and shelves into people's arms, panelists said.
"Those factors, along with consistent, fact-based communications, will help us begin to build up the public’s confidence in these vaccines and in vaccination," Moore said.
So far, the government under President Donald Trump has delivered more than 31 million doses of the two vaccines across America. But only 12 million – or about 39% – have been used.
What's needed, the panelists said, are mass vaccination clinics that together with other distribution methods can deliver more than 1 million shots a day. Biden has promised 100 million shots will be given during his first 100 days in office, which starts Wednesday.
A national scheduling tool would be a first step, according to several panel members, including Prashant Yadav, a medical supply chain expert and senior fellow with the Center for Global Development, an international development think tank based in Washington, D.C., and London.
Disneyland knows how to schedule to maximize riders and minimize lines by giving people hourly arrival windows instead of precise times, he said. Customers waiting for bank tellers join a common line, which moves faster than five separate ones.
And airlines prioritize seating to keep first-class passengers happy, he noted. Why can't the same be done for people who should be prioritized to receive vaccinations?
Right now, every state is on its own to figure out how best to schedule. But, Yadav said, “providing a scheduling software is a thing we should do federally, so we have economies of scale and scope – in communicating about it, in training people to use it and in getting the data back."
More funding for pandemic-weary state and local health departments also is essential, according to the panel. Although Congress allocated $3 billion in December for vaccine distribution, that money has yet to arrive in states, and when it does, it will take time to make a difference.
The 15 USA TODAY panelists, who have expertise in everything from virology to logistics, remain optimistic the vast majority of Americans who want a vaccine will be able to get one by summer.
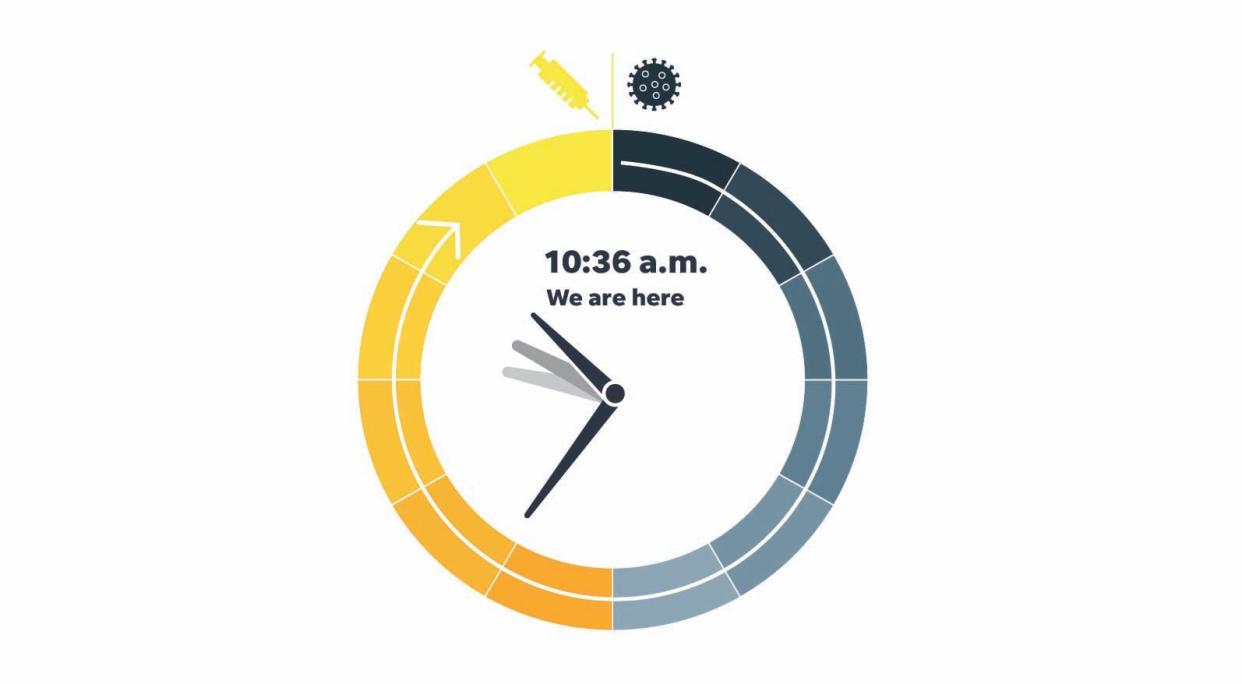
Every month since June, we have asked panelists what time it is on an imaginary clock that started ticking at midnight a year ago, when people were first infected with the new coronavirus. It will reach high noon when vaccine is widely available to all Americans. Each month, we calculate the median time – the midpoint of their estimates.
Starting at 4 a.m., the clock moved forward an hour every month, except November, when it jumped ahead 90 minutes, buoyed by the results of the first two vaccine trials. This month, the clock barely budged, advancing only 6 minutes, from 10:30 a.m. to 10:36 a.m.
Speeding up vaccinations
Some of the panelists envision mass vaccination campaigns with most people scheduled to keep lines moving and vaccinators busy.
“We need high throughput,” said Dr. Monica Gandhi, a professor of medicine and infectious disease expert at the University of California-San Francisco.
Volunteers without medical training can log people in before passing them to others who can administer consent forms. Then, trained medical personnel can deliver the shots, while a final medical group watches people for 15 minutes after vaccination to make sure they don't have adverse reactions.
To get as many people vaccinated as possible, the vaccinators should never be waiting, Yadav said.
"The scarcest resource in the system is the vaccinator. This system doesn’t let the vaccinators’ time go idle,” he said.
Gandhi said there should be online reservations, but also drop-in slots. "Because of the digital divide and language barriers you need both,” she said.
Such sites should be in addition to shots given at doctors' offices, clinics pharmacies and local health centers. But the mass vaccination centers would allow large numbers of people to get immunized even while smaller settings reach those more reticent about the vaccine or live outside population centers, said Gandhi.
Nursing and medical students can be conscripted into the effort, along with other nontraditional health-care workers, said Prakash Nagarkatti, vice president for research at the University of South Carolina.
"All states should have engaged such volunteers several months before the vaccine rollout," he said.
While several panelists praised the Trump administration for getting two vaccines to the finish line, with three more closing in, they criticized the lack of foresight to get vaccine from loading docks into arms.
"There was total lack of planning at the state level for mass vaccination and the federal government did not help the states overcome the hurdles," Nagarkatti said.
"We spent so much time on science and manufacturing and politics and we completely dropped the ball on logistics," said Peter Pitts, president and co-founder of the Center for Medicine in the Public Interest, a nonprofit research group.
It's something the United States has never been good at, said Arti Rai, a law professor and health law expert at Duke University Law School.
"Since the days of Vannevar Bush in World War II, the U.S. has done very well on the life sciences R&D side, somewhat less well on the manufacturing side, and poorly on the fragmented delivery side," she said.
Beefing up communication
Pitts said he's seen a shift in public attitudes in recent months from people dismissing the "Trump vaccine" to asking when and where they can get their shot.
"People understand both of these vaccines have been approved by the FDA without any shenanigans," he said. "People are actively excited and anxious to get vaccinated."
But Pitts has been incredibly frustrated by the lack of public education. With more than $10 billion spent on developing, manufacturing and distributing the vaccines at the federal level, so far little money has gone to communicating about when, where and why people should get vaccinated.
The Department of Health and Human Services has promised a long-delayed education campaign aimed at encouraging Americans to get vaccinated will launch in January but no date has been set.
Federal officials underestimated vaccine hesitancy and have failed to address many of the public's questions, said Dr. William Schaffner, a professor and infectious disease expert at the Vanderbilt University School of Medicine in Nashville, Tennessee.
Schaffner said his own hospital conducted surveys, then launched an education campaign to help people understand the vaccine's record and development process.
“We had Q&As for all different categories of employees who work at this medical center," he said via email. "We actually saw the needle moving among all sectors."
Vanderbilt has given initial shots to 18,000 employees so far, he said, and is now working on getting them the required second dose, while encouraging even more to get vaccinated.
One group that has remained hesitant, he said, are young women, who fear – despite no evidence or biological plausibility – that the vaccine could affect their fertility.
"There’s a lot of nonsense on the Internet having to do with sterility and reproductive health and the vaccine," he said. "We did a video specifically on that topic for staff.”
Changing some people's behavior will have a ripple effect, Schaffner said. “They look left and right to see what their peers are doing."
The failure to launch such education campaigns has real-world consequences, Pitts said.
Without an active effort to encourage vaccination among Black and Hispanic health care workers, many declined the vaccine when they had an early chance for a shot.
So now, instead of serving as the role models they could have been for others in their communities, "we have respected health care members of communities of color saying 'Don't get vaccinated,'" Pitts said. "Not only did we not engage them to do the right thing, they're actually out there now hurting the effort."
Time for a rollout reset
All the panelists said they are looking forward to a new administration.
"The Biden administration is serious about ending the epidemic, and therefore they will certainly make a concerted effort to ensure mass vaccination," Nagarkatti said, reflecting the group's view.
Dr. Paul Offit, director of the Vaccine Education Center at Children's Hospital of Philadelphia, said the rollout will get smoother with time and calmer under the new administration. "At least we won't be getting proclamations of disdain for every science-based federal agency," he said.
He's also looking forward to the arrival of spring and summer, when he believes enough people will be vaccinated to make a difference, and the infection rate will naturally decline for a few months.
"All these good things are about to happen," Offit said. "We will get a more humid climate. Good. We have an administration that has a clue. Good. We have a vaccine that works really well. Good. And, the best part is it's not going to be just these two vaccines."
Two more vaccines are nearing completion of their clinical trials and will likely be authorized for use in February and March, Offit said, which "lightens the load in terms of production."
Several panelists also had suggestions for what the Biden administration should do to improve the vaccination campaign.
“Biden should create this new cadre of operations people who can help every state," Yadav said. "If Florida says they don’t want it, then OK. But if a state wants it, we should be giving it to them.”
Vivian Riefberg, professor of practice at the Darden School of Business at the University of Virginia, said the new administration should enlist as many approaches to vaccination as possible: mass distribution centers, community health facilities, physician's offices, pharmacies, schools.
"You've got to provide an array of places that administer vaccines," she said.
Coordination and communication must be improved between the federal and state governments.
A better information technology system at the national level can help with registration, enrollment, and tracking, she said.
Right now, state systems are crashing because they are overloaded with people trying to sign up, leaving some on hold for hours or having to call dozens of times to get through. This disadvantages people who are likely to be most in need of vaccination.
Finally, Riefberg said, the new administration needs to address resistance to vaccination through education campaigns, enlisting trusted role models, even paying people to administer and receive shots, if necessary.
But she cautioned against expecting too much too quickly.
"We need to give the Biden people a chance to get off the ground here," she said, "especially given the array of changes that are happening right now."
How we did it
USA TODAY asked scientists, researchers and other experts how far they think the vaccine development effort has progressed since Jan. 1, when the virus was first recognized. Fifteen responded. We aggregated their responses and calculated the median, the midway point among them.
This month's panelists
Pamela Bjorkman, structural biologist at the California Institute of Technology
Dr. Monica Gandhi, an infectious disease expert at the University of California, San Francisco
Sam Halabi, professor of law, University of Missouri; scholar at the O’Neill Institute for National and Global Health Law at Georgetown University
Florian Krammer, virologist at the Icahn School of Medicine at Mount Sinai in New York City
Dr. Kelly Moore, deputy director of the non-profit Immunization Action Coalition; former member of the CDC Advisory Committee on Immunization Practices; chair, World Health Organization Immunization Practices Advisory Committee
Prakash Nagarkatti, immunologist and vice president for research, University of South Carolina
Dr. Paul Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children's Hospital of Philadelphia and a professor of Vaccinology at the Perelman School of Medicine at the University of Pennsylvania
Peter Pitts, president and co-founder of the Center for Medicine in the Public Interest, and a former FDA Associate Commissioner for External Relations
Dr. Gregory Poland, director, Mayo Clinic's Vaccine Research Group, and editor-in-chief, Vaccine
Arti Rai, law professor and health law expert at Duke University Law School
Vivian Riefberg, professor of practice at the Darden School of Business at the University of Virginia, director emeritus and senior advisor with McKinsey & Company, and a board member of Johns Hopkins Medicine, PBS, and Signify Health, a healthcare platform company working to transform how care is paid for and delivered at home.
Erica Ollmann Saphire, structural biologist and professor at La Jolla Institute for Immunology
Dr. William Schaffner, a professor and infectious disease expert at the Vanderbilt University School of Medicine in Nashville, Tennessee.
Prashant Yadav, senior fellow, Center for Global Development, medical supply chain expert
Dr. Otto Yang, professor of medicine and associate chief of infectious disease at the David Geffen School of Medicine at UCLA
Contact Karen Weintraub at kweintraub@usatoday.com and Elizabeth Weise at eweise@usatoday.com.
Health and patient safety coverage at USA TODAY is made possible in part by a grant from the Masimo Foundation for Ethics, Innovation and Competition in Healthcare. The Masimo Foundation does not provide editorial input.
This article originally appeared on USA TODAY: Experts slam COVID-19 vaccination rollout, look forward to Biden

 Yahoo Movies
Yahoo Movies 
