Clinical trials need more Black and Latino people. Everyone should get paid to participate.
When the Supreme Court ruled against the NCAA and in favor of compensating college athletes, I considered how this strategy could help solve the lack of diversity in clinical research and clinical trial enrollment. The two topics may seem unrelated, but they're strikingly similar. Collegiate athletes from low-income or working-class families often live financially strapped lives on campus. Even those who are compensated receive relatively small stipends that are a fraction of what they earn for their schools.
The high court decision addressed persistent inequities in college athletics. It acknowledged that a full scholarship and stipend may not be enough for some students, who could otherwise work a part-time job but are unable to because of their academic and athletic commitments. It also addressed how colleges reap enormous profits from a student's image, name, and likeness while forbidding student-athletes to do the same.
Compensation boosts participation
Similarly, the difficulties of assuming an often overlooked financial burden are at work in clinical trial recruitment. The inability to take on the costs of participating in a trial is one of the primary reasons why trial diversity remains an intractable problem.
People of color are barely represented in trials for conditions that impact them in far greater numbers than white people, including diabetes and cancer. The most recent multi-year report from the Food and Drug Administration found only 7% of clinical trial participants were Black and only 13% were Hispanics. In heart disease trials, 3% of participants were Black men. These shares are abysmal and haven't changed in years, despite prolonged discussion and attempts to fix the problem that haven't made an appreciable difference.
Black medical leaders: Coronavirus magnifies racial inequities, with deadly consequences
I spent nearly five years at the FDA's Center for Drug Evaluation and Research working to increase diversity in clinical trials. Increasing the number of enrollment sites has been done and hasn't worked. Relying on digital advertising and community partnerships has had limited success. It's time to recognize that other issues are at play, and one of them is the cost of participating.
Some clinical trials do reimburse for some costs associated with trial participation. But the process is disjointed, and reimbursement levels are determined by drug and device companies sponsoring the research. An independent review board, or IRB, then reviews the decision on payment and has the final say in how much a participant will receive, if anything, for each study.
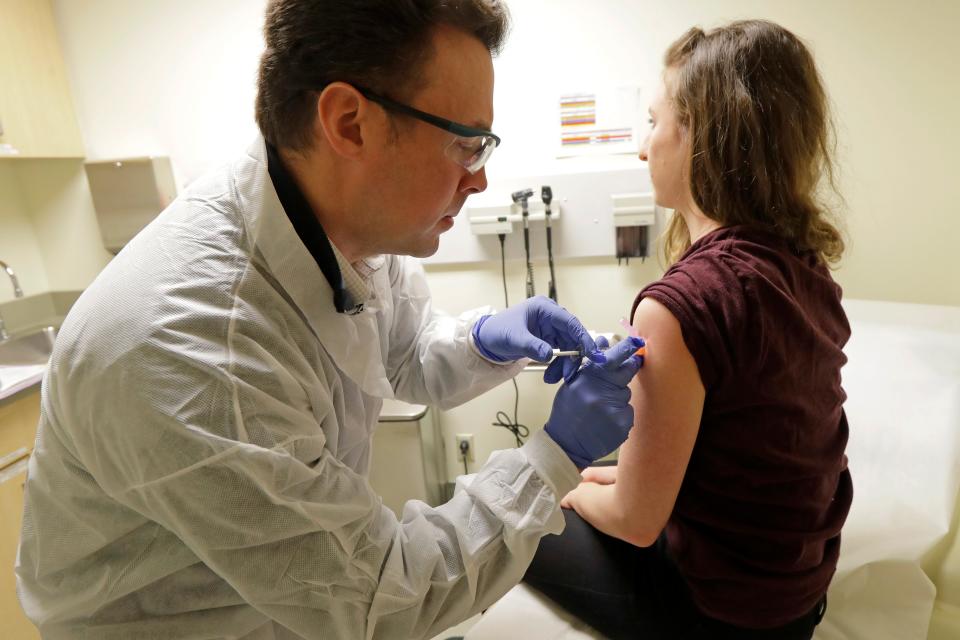
The pay range for participation can vary widely, with an average of about $50 to $300 per day, but some trials don't pay anything.
Patients travel hundreds of miles to enroll in clinical trialshat can save or extend their lives. Other financial hurdles that prevent enrollment in trials include parking, taking time off work, and child care or elder care. Researchers, clinical trial sites and pharmaceutical companies need to stop turning a blind eye to these significant barriers. They need to design trials that support participants in managing these challenges and compensate them for all routine costs of participating – from travel costs to lost time from work.
For too long, the FDA has taken a paternalistic approach to protecting patients from "unethical inducements" to ensure they won't be taken advantage of or coerced to participate. But the argument is disingenuous. The reality is that if enrollees aren't compensated for lost wages, travel, caregiving costs and any co-pays and deductibles that are not considered a routine trial cost by an insurer, then unfairness is already at work. There is nothing ethical about it.
Those who give most benefit least
Pharmaceutical companies, universities, researchers and clinical trial sites all profit from the contributions of clinical trial participants, who literally give of themselves through blood, skin and tissue samples. Just like in college sports, everyone is benefitting except the people giving the most.
Let's not presume that the benefit to patients should simply be the ability to participate. It's unrealistic to believe that people are going to enter clinical trials – often experimental medicines and invasive procedures – to make money. Patients don't enroll in trials to get paid. They enroll to get better and live longer. When we don't pay patients to participate in clinical trials, we limit enrollment.
Insurance executives: Want to reduce health care inequities? Lessons from our COVID vaccine campaign
The Supreme Court decision sought to rectify an inequity in collegiate sports. Similarly, we need a uniform, regulated approach to clinical trial compensation that reflects the value of a person's commitment to research and the impact of a person's enrollment on their economic experience.
We desperately need to improve diversity in clinical trials. Paying people to participate will not only compensate them for financial losses, but create a more representative sample for new treatments.
John Whyte, MD, MPH, is chief medical officer of WebMD. Follow him on Twitter: @DrJohnWhyte
You can read diverse opinions from our Board of Contributors and other writers on the Opinion front page, on Twitter @usatodayopinion and in our daily Opinion newsletter. To respond to a column, submit a comment to letters@usatoday.com.
This article originally appeared on USA TODAY: Improve health equity, diversity and clinical trials: Pay participants

 Yahoo Movies
Yahoo Movies 
